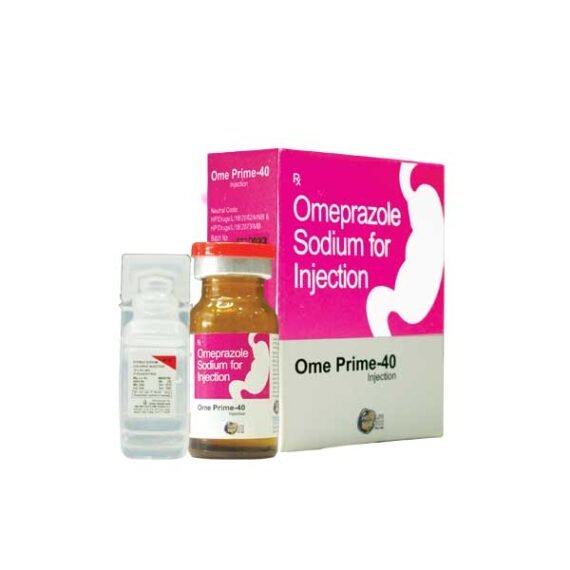
Ome Prime-40 Injection
$0.00$0.00
Brand:- Ome-Prime 40
Composition:-Omeprazole Sodium for Injection
Form:- Injection
Strength:- 40mg
Packing:- 1 Vial Injection
Manufacture By:- Prime Life Sciences Pvt Ltd
DESCRIPTION:
Omeprazole, a racemic mixture of two enantiomers reduces gastric acid secretion through a highly targeted mechanism of action. It is a specific inhibitor of the acid pump in the parietal cell. It is rapidly acting and provides control through reversible inhibition of gastric acid secretion with once-daily dosing.
Description
Description
What does the Ome-Prime 40 contain?
Each vial of powder for solution for injection contains omeprazole sodium 42.6 mg, equivalent to omeprazole 40 mg. After reconstitution, 1 ml contains omeprazole sodium 4.26 mg, equivalent to omeprazole 4 mg. For a full list of excipients, see section 6.1.
How to take a Ome-Prime 40?
- Higher daily doses may be required and the dose should be adjusted individually.
- When doses exceed 60 mg daily, the dose should be divided and given twice daily. Omeprazole is to be administered in an intravenous infusion for 20-30 minutes.
Possible side effects of Ome-Prime 40.
The most common adverse events (1-10% of patients) are headache, abdominal pain, constipation, diarrhoea, flatulence, and nausea/vomiting. The following adverse reactions have been identified or suspected in the clinical trials programme for omeprazole and post-marketing.
Storage.
Do not store above 25 °C. Keep the vial in the outer container.


























Reviews
There are no reviews yet.